Sodium polyacrylate and water

Sodium polyacrylate can absorb over 200 times its mass in water.
Fluorine reacting with hydrogen in a balloon

A neodymium magnet falling through a copper pipe

Magnetic field equilibrium.
A kernel of corn popping


You can also do this (to a lesser extent) by rubbing a balloon on your hair, then holding it next to your running sink.
Mercury(II) thiocyanate decomposition

Heating mercury(II) thiocyanate causes it to release toxic gases and decompose into an insoluble brown mass.
Hydrogen peroxide mixed with potassium iodide

The iodide catalyzes the decomposition of the hydrogen peroxide. A small amount of dishwashing soap is usually added so that the resultant oxygen is caught in the bubbles, forming a tower of foam.
Copper reacting with nitric acid

Oxidation.
Electrical treeing

Electrical discharge.

Firework explosion in slow motion

Belousov–Zhabotinsky reaction
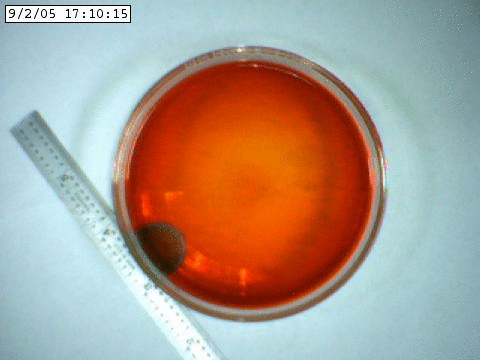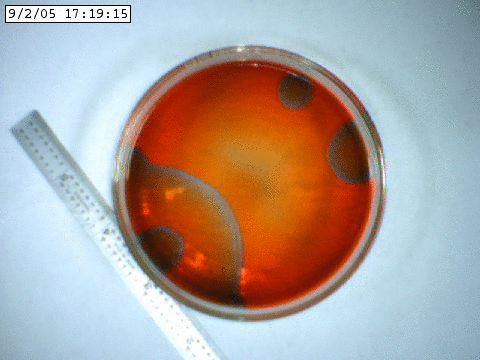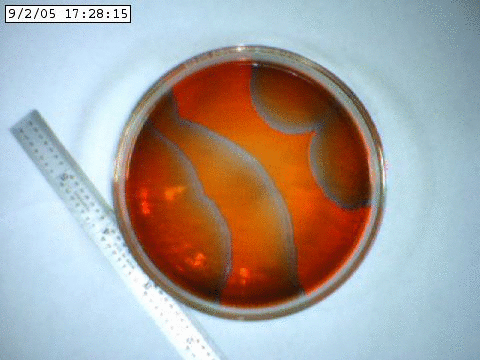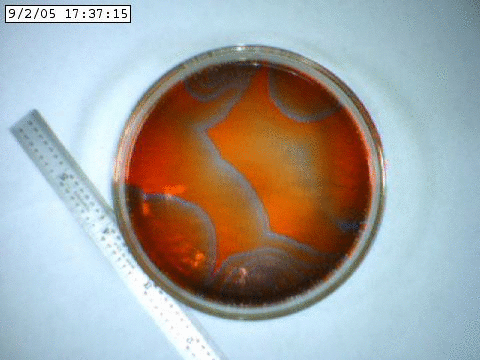
A slowly propagating chemical oscillation containing bromine and an acid.
Lithium on fire

A candle lit by its vapor trail

Orange LED light immersed in liquid nitrogen

The wavelength & frequency of the light changes as it loses thermal energy.
Flammable fluid in a glass jug

Isopropyl alcohol, most likely.
 Barnorama All Fun In The Barn
Barnorama All Fun In The Barn